[vc_row][vc_column][vc_column_text]
Nerve cell membranes are the communication centers of the brain. It is through membrane processes that nerve cells communicate with one another and also turn on/off or up/down regulate cell genome activity. In addition, membranes allow the organized and controlled movement of nutrients into and waste products out of nerve cells. Many of these membrane functions require the input of cellular metabolic energy which accounts for nerve cell membranes, especially synaptic membranes, being the highest consumers of metabolic energy in the brain.
Membranes are composed of two major classes of molecules – lipids and proteins – with phospholipids and cholesterol being the major lipids (Model Membrane). Membrane proteins give rise to molecular clusters which function as membrane receptors, channels, and enzymes which convert proteins and lipids into smaller or larger structures. Membrane proteins are embedded in the membrane lipids. Therefore, alterations in the structure and molecular dynamics of the membrane lipids will potentially alter the dynamics, structure, and physiological function of the membrane proteins. We recently demonstrated that by altering the structure and dynamics of a model membrane, the structure and dynamics of the embedded protein was clearly altered (Mandal and Pettegrew, 2004). In addition, a recent study demonstrated that the molecular dynamics of a protein were important for its normal enzymatic function (Eisenmesser et al., 2005; Haung and Montelione, 2005).
For many decades it was assumed that membrane proteins were the important players and lipids had only supportive, secondary roles. However over the last 30 years, Dr. Pettegrew has demonstrated that lipids are equally if not more important in normal membrane function than proteins. In addition, Dr. Pettegrew has demonstrated that the molecular dynamics of membrane phospholipids as well as membrane lipid composition and turnover are clearly altered in diseases such as Alzheimer’s disease, autism, chronic alcoholism, major depressive disorder and schizophrenia. These studies demonstrate that alterations in membrane lipid turnover and composition can lead to alterations in membrane protein structure and dynamic motion. Therefore, altering the type and numbers of lipids on the “molecular dance floor” will alter the ability of proteins to interact and “dance with each other” which is vitally important for their normal physiological function.
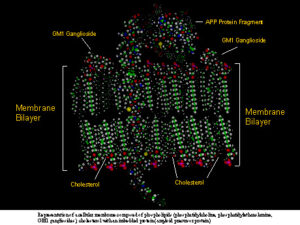
(PtdEtn)], GM1 ganglioslide, and cholesterol with an imbedded protein fragment [amyloid precursor protein (APP) fragment].
[/vc_column_text][/vc_column][/vc_row][vc_row][vc_column][penci_text_block block_title_align=”style-title-left” custom_markup_1=””]
Relevant Articles
| Eisenmesser EZ, Millet O, Labeikovsky W, Korzhnev DM, Wolf-Watz M, Bosco DA, Skalicky JJ, Kay LE and Kern D. (2005) Intrinsic dynamics of an enzyme underlies catalysis. Nature 438, 117-121.
Haung YJ and Montelione GT. (2005) Structural Biology: Proteins flex to function. Nature 438, 36-37. Mandal PK and Pettegrew JW. (2004) Alzheimer’s disease: soluble oligomeric Αβ(1-40) peptide in membrane mimic environment from solution NMR and CD studies. Neurochemical Research, 29, 2255-2260. |
[/penci_text_block][/vc_column][/vc_row]